

History
2025
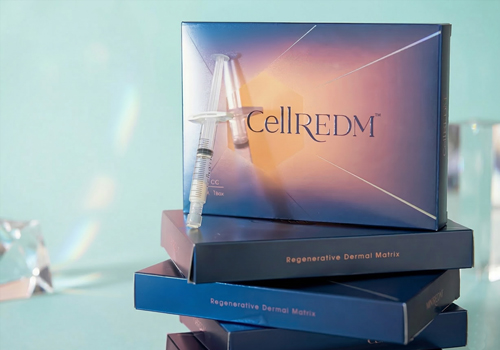
2025
·Launch of the ECM Collagen Booster, CellREDM™
2024
·Launch of "Elatense", a tightening band for double chin and neck wrinkle improvement
·Approval of 3 MINT Lift products by the Thai TFDA
·Approval of 7 MINT Lift products by the Chinese NMPA
·Signed MOU with WEGO Group for China’s human tissue and medical device market
·Launched two new skin boosters under HansPharma
·Signed second technology transfer agreement for Korean genome chip v2.0 with Korea National Institute of Health
·MINT Lift awarded CSBA Consumer Satisfaction Brand for 5 consecutive years
·Launch of allogenic dermal collagen filler "BellaGen Plus"
·Launch of two allogenic bone grafts for periodontal use
·Selected as a family-friendly company
·Approval of 3 MINT Lift products by the Thai TFDA
·Approval of 7 MINT Lift products by the Chinese NMPA
·Signed MOU with WEGO Group for China’s human tissue and medical device market
·Launched two new skin boosters under HansPharma
·Signed second technology transfer agreement for Korean genome chip v2.0 with Korea National Institute of Health
·MINT Lift awarded CSBA Consumer Satisfaction Brand for 5 consecutive years
·Launch of allogenic dermal collagen filler "BellaGen Plus"
·Launch of two allogenic bone grafts for periodontal use
·Selected as a family-friendly company


2023
·MINT Lift selected as World-Class Product
·Signed supply contract with Osstem Implant for Chinese market
·Signed MOU with KTC to enhance collaboration in bio-health and medical device technology
·Signed supply contract with Osstem Implant for Chinese market
·Signed MOU with KTC to enhance collaboration in bio-health and medical device technology
2022
·Launched "Kladie Cell-up Solution" by HansPharma
·Signed research agreement with MINT Hospital on NK cell development
·Signed MOU with Korea Public Tissue Bank for human tissue cooperation
·Signed research agreement with MINT Hospital on NK cell development
·Signed MOU with Korea Public Tissue Bank for human tissue cooperation


2021
·Established JV "LEVOS" with WEGO Group
·MINT Lift won Excellent Brand Award for two consecutive years
·Declaration of Ethical Management
·MINT Lift won Excellent Brand Award for two consecutive years
·Declaration of Ethical Management
2019
Achieved the largest market share in the domestic artificial breast implant market
Completed the construction of the first artificial breast implant automation facility (smart factory) in the world
Established Hans Biomed U.K
Established Hans Biomed Mexico
Constructed a high-tech medical equipment research center
Completed the construction of the first artificial breast implant automation facility (smart factory) in the world
Established Hans Biomed U.K
Established Hans Biomed Mexico
Constructed a high-tech medical equipment research center


2018
Signed a contract on the transfer of neural stem cell culture technology with the College of Medicine, Yonsei University
Acquired BNA Med, the sales business of the domestic plastic surgery clinic
Merged Cosmeceutical Zitones Bio and Hans Pharma
Established Hans Care, a sales business for the domestic plastic clinics
Established Hans Biomed Thailand
Acquired BNA Med, the sales business of the domestic plastic surgery clinic
Merged Cosmeceutical Zitones Bio and Hans Pharma
Established Hans Care, a sales business for the domestic plastic clinics
Established Hans Biomed Thailand
2017
Constructed Munjeong new office building (Seoul) of Hand Biomed Co/. Ltd.
Licensed to manufacture and sell BellaGel SmoothFine by MFDS
Licensed to manufacture and sell MINT Easy & Fine by MFDS
Constructed a research center for cell therapy products (Seoul)
Licensed to manufacture and sell BellaGel SmoothFine by MFDS
Licensed to manufacture and sell MINT Easy & Fine by MFDS
Constructed a research center for cell therapy products (Seoul)

2016
Licensed to sell “BellaGel” (artificial breast implant) from Chinese National Medical Products Administration (CNMPA)
2015
Licensed to manufacture and sell artificial breast implants 'BellaGel' by MFDS
Established “Hans Pharma” a subsidiary of Hans Biomed Co. in the field of cosmeceutical market.
Established Shanghai Hans Biomed Corp.
Established “Hans Pharma” a subsidiary of Hans Biomed Co. in the field of cosmeceutical market.
Established Shanghai Hans Biomed Corp.


2014
Licensed to manufacture and sell Mint Lift facial tissue fixing sutures by MFDS
Awarded "$10 Million Export Tower” on the 51st Trade Day
Licensed to manufacture and sell XenoDerm by MFDS
Licensed to manufacture and sell Scar Clinic (gel-type scare care product) by MFDS
Awarded "$10 Million Export Tower” on the 51st Trade Day
Licensed to manufacture and sell XenoDerm by MFDS
Licensed to manufacture and sell Scar Clinic (gel-type scare care product) by MFDS
2013
Extended Hans Daeduk Research Center (the tissue engineering research lab)
Awarded "$5 Million Export Tower" on the 20th Trade Day
Awarded "$5 Million Export Tower" on the 20th Trade Day
2011
Established Hans Biomed USA Inc.

2009
Listed on KOSDAQ, the first in the industry
2008
Artificial breast registered for CE
Awarded "$ 1Million Export Tower" (KTA)
Awarded "$ 1Million Export Tower" (KTA)
2006
Commercialized DBM (Demineralized Bone Matrix)
(Product Name: SuerFuseTM-Gel/Putty, ExFuseTM-Gel/Putty)
(Product Name: SuerFuseTM-Gel/Putty, ExFuseTM-Gel/Putty)
2005
Acquired permission for establishing the first 'tissue bank' in Korea from MFDS


2004
Launched “Scar Clinic” (a scar care product) first in Korea
2003
Registered a human tissue transplant to the U.S. FDA

2002
Constructed Hans Deduk Research Center (tissue engineering research lab) on a total floor area of 2,644m2 (as of now, 5.950m2)
2001
Established Japan Hans Biomed Co. Ltd.

2000
Developed SuerDerm TM (Acellular Dermal Matrix)
Established HITE Hans Institute of Tissue Engineering
Established HITE Hans Institute of Tissue Engineering
1999
Established Hans Medical Co., Ltd.
Researched and developed acellular dermis with KAIST
Researched and developed acellular dermis with KAIST
1993~1998
Produced compression clothing exclusive for burn treatment and liposuction surgery


-
product
- Biologics
- Medical Device
-
recruit
- Job Opening
- Application
- HR System